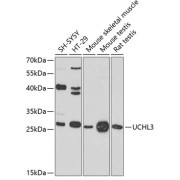
Ubiquitin Carboxyl-Terminal Hydrolase Isozyme L3 (UCHL3) Antibody

292.5€ (80 µl)
Por favor contáctenos para obtener información detallada sobre el precio y disponibilidad.
Name
Ubiquitin Carboxyl-Terminal Hydrolase Isozyme L3 (UCHL3) Antibody
Category
Primary Antibodies
Provider
Abbexa
Reference
abx031536
Tested Applications
ELISA, WB, IHC
Description
Covalent attachment of the C-terminus of ubiquitin to cellular proteins plays a role in a variety of cellular processes. Ubiquitin C-terminal hydrolysis is catalyzed by deubiquitinating (DUB) enzymes and is necessary for several functions, including liberation of monomeric ubiquitin from the precursors encoded by ubiquitin genes and recycling of ubiquitin monomers. There are 2 distinct families of DUBs, ubiquitin-specific proteases (UBPs) and ubiquitin C-terminal hydrolases (UCHs). Mayer and Wilkinson (1989) identified 4 distinct UCH activities from bovine thymus. All 4 were thiol proteases and had high-affinity binding sites for ubiquitin. Wilkinson et al. (1989) purified the predominant isozyme, UCHL3, and raised antibodies against it. By screening a human B-cell expression library with the antibodies, the authors isolated cDNAs encoding human UCHL3. Sequence comparisons revealed that the sequence of the predicted 230-amino acid human UCHL3 protein is 54% identical to that of UCHL1.
Documentos del producto
Instrucciones
Data sheet
Especificaciones del producto
| Category | Primary Antibodies |
| Immunogen Target | Target: Ubiquitin Carboxyl-Terminal Hydrolase Isozyme L3 (UCHL3) Immunogen: KLH-conjugated synthetic peptide between 195-225 amino acids from the C-terminal region of human UCHL3. |
| Host | Rabbit |
| Reactivity | Human |
| Recommended Dilution | WB: 1/1000, IHC-P: 1/10 - 1/50. Not tested in IHC-F. Optimal dilutions/concentrations should be determined by the end user. |
| Clonality | Polyclonal |
| Conjugation | Unconjugated |
| Isotype | IgG |
| Observed MW | Calculated MW: 26.2 kDa |
| Purification | Purified through a protein G column, eluted with high and low pH buffers and neutralized immediately, followed by dialysis against PBS. |
| Size 1 | 80 µl |
| Size 2 | 400 µl |
| Form | Liquid |
| Tested Applications | ELISA, WB, IHC |
| Buffer | PBS containing 0.09% sodium azide. |
| Availability | Shipped within 5-10 working days. |
| Storage | Aliquot and store at -20°C. Avoid repeated freeze/thaw cycles. |
| Dry Ice | No |
| UniProt ID | P15374 |
| NCBI Accession | NP_001257881.1, NP_005993.1 |
| Alias | UCH-L3,Ubiquitin thioesterase L3 |
| Background | Antibody anti-UCHL3 |
| Status | RUO |
| Note | THIS PRODUCT IS FOR RESEARCH USE ONLY. NOT FOR USE IN DIAGNOSTIC, THERAPEUTIC OR COSMETIC PROCEDURES. NOT FOR HUMAN OR ANIMAL CONSUMPTION. |
Background
UCHL3 is a deubiquitinating enzyme involved in ubiquitin recycling and protein turnover, ensuring cellular protein quality control and homeostasis. It cleaves ubiquitin precursors and removes ubiquitin chains from target proteins, preventing their premature degradation by the proteasome. UCHL3 is ubiquitously expressed and plays roles in DNA repair, apoptosis, and embryonic development. It is particularly essential for spermatogenesis, where it regulates chromatin remodeling and germ cell differentiation. Loss of UCHL3 function is associated with impaired cellular proteostasis, genomic instability, and reproductive defects. Studies show its involvement in tumor progression, where dysregulation of UCHL3 alters protein degradation pathways, contributing to cancer cell survival. Knockout models exhibit defects in germ cell development, growth retardation, and increased sensitivity to cellular stress, underscoring its importance in ubiquitin regulation, cellular stability, and reproductive biology.
Productos relacionados
Ubiquitin Carboxyl-Terminal Hydrolase Isozyme L3 (UCHL3) Antibody
UCHL3 Antibody is a Rabbit Polyclonal Antibody against UCHL3.…
Ver producto
Ubiquitin Carboxyl-Terminal Hydrolase Isozyme L3 (UCHL3) Antibody
Covalent attachment of the C-terminus of ubiquitin to cellular proteins plays a role in a variety of cellular processes.…
Ver producto
Human Ubiquitin Carboxyl Terminal Hydrolase L3 (UCHL3) Protein
Human Ubiquitin Carboxyl Terminal Hydrolase L3 (UCHL3) is a recombinant Human protein expressed in E. coli.…
Ver producto